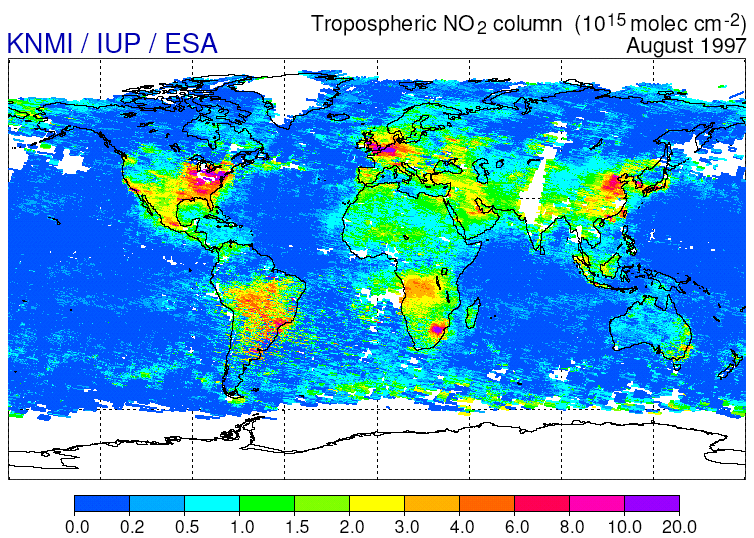
Univ. of Arizona (credits)
よくある質問は、なぜ二酸化窒素 (NO 2 )、二酸化硫黄 (SO 2 )、および一酸化炭素 (CO) の濃度が常に低く、それらの濃度を測定することが役立つのですか?というものです。
これらの物質を監視することの重要性を強調するために、この記事では、これらの汚染物質がどこから来て、私たちの健康や環境にどのような影響を与えるのかについて説明します。
話を短くするために、この記事では NO2 だけに焦点を当てます。
--
二酸化窒素 (NO 2 ) とは何ですか?
NO 2は、すべての燃焼エンジンから排出される赤茶色のガスです。燃焼エンジンから排出される窒素ベースの主な化合物は、NO 2と一酸化窒素 (NO) の 2 つです。これら 2 つの汚染物質を総称して、NO xまたは窒素酸化物と呼びます。
要約する:
- NO2: Nitrogen Dioxyde
- NO: Nitric Dioxyde
- NOx: Oxides of Nitrogen = {NO2+NO}
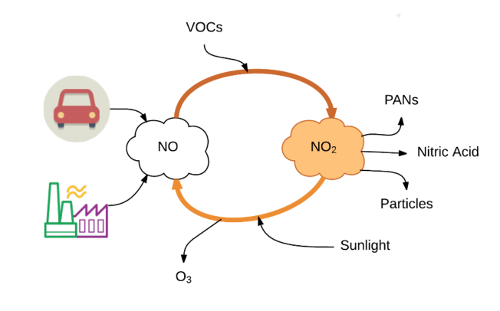
NO 2 のライフサイクル
排出点 (つまり、排気管) では、NO xの割合は約 90% が NO、10% が NO 2 (1) です。
大気中で揮発性有機化合物 (VOC) の存在下で数時間放置すると、NO は NO 2に変換されます。この反応は、数秒から数時間かけて起こることがあります (2)。
NO 2は空気中の他の物質とさらに反応して、硝酸、粒子状物質、および PAN (硝酸ペルオキシアシル) と呼ばれる物質を形成します。
また、太陽光によって NO 2 がNO に戻り、副次的な汚染物質としてオゾン (O 3 ) が生成される可能性があります。 NO 2はこれらの「二次」汚染物質を生成する可能性があるため、NO 2を監視し規制することが重要です。
NO2は私にどのような影響を与えますか?
NO 2への短期および長期の曝露は、呼吸器疾患のリスク増加と関連しています。喘息患者、幼児、高齢者は喘息の影響に対する感受性が高くなります(1)。
大気中の NO 2の存在によって引き起こされる二次汚染物質にも、それ自体の悪影響があります。 PAN は刺激物であり、硝酸は酸性雨や粒子状物質を引き起こし、O3 は呼吸器疾患を引き起こします。
誰が、何がNOxを排出しているのでしょうか?
前述したように、NO x排出は燃焼エンジンから発生します。しかし、これらのエンジンで起こっているのは、化石燃料中の炭化水素と空気 (80% が窒素) との高温反応です。山火事や落雷などの天然の NO x排出源もありますが、大気中の NO 2の大部分は人間の活動によって引き起こされます。
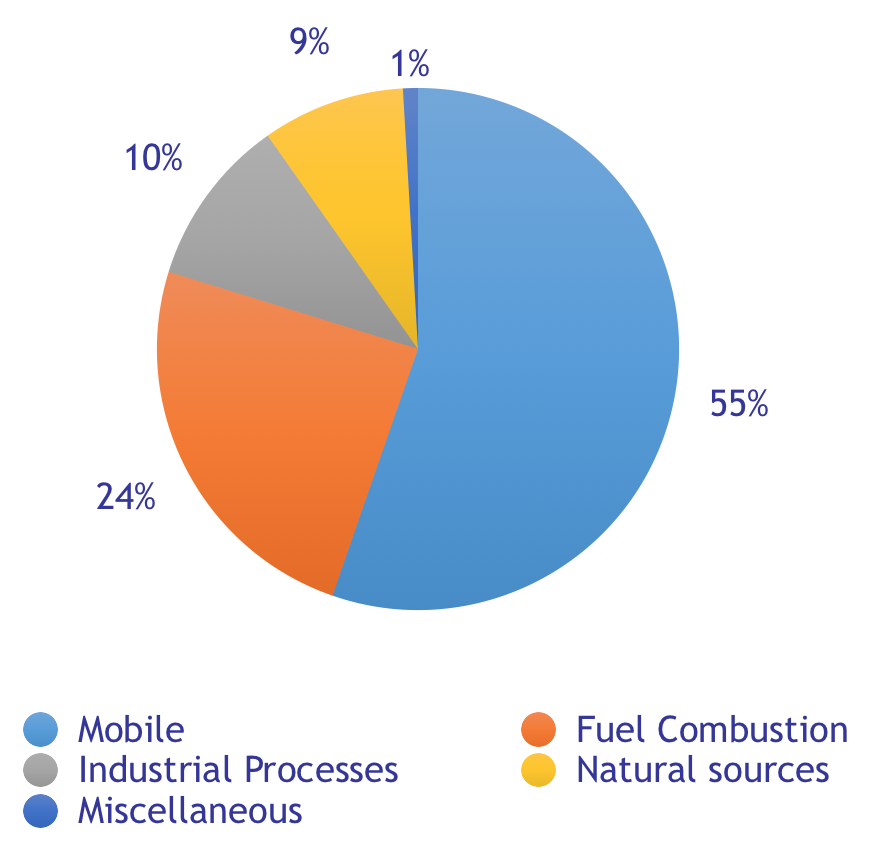
NO xを排出する産業は数多くあります。図 (2) は、2014 年に測定された米国の NO x排出源を示しています。 「移動発生源」 、つまり道路車両、ボート、飛行機、農用車などは、間違いなく最大の排出源です。米国とヨーロッパでは、道路車両が NO xの主な「移動発生源」です (1) (3)。
道路車両が NO xの主な発生源である地域では、以下の NO 2 AQI に示すように、平日の午後 5 時頃などの交通のピーク時間帯に NO 2濃度が高くなることがよくあります。追加の興味深いメモ:
- オゾン(O 3 )濃度は日中増加し、NO 2濃度は減少します。それは、NO 2が太陽光の存在下で NO xに変化するためです。
- 午後 6 時を過ぎると、NO 2 を NO に戻す太陽光がなくなるため、NO 2濃度が増加します。

Figure 3 Example of AQI in London
しかし、中国では 2013 年に実施された調査で、産業発生源が NO xの最大 (全体の 34%) 排出者であることが判明しました (4)。これが、NO 2濃度とラッシュアワーの交通との間に強い関連性がない理由です。
なぜNO 2濃度はこれほど低いのでしょうか?
NO 2 AQI は通常「良好」の範囲にあり、緑色で表示されます。しかし、NO 2はそれ自体に悪影響を与える他の汚染物質に変換されるため、大気中の NO 2 は依然として私たちの健康と環境に悪影響を及ぼします。大気中の NO xについてさらに詳しい説明が必要な場合は、以下の文書を参照してください。
参考文献と詳細情報
2. Cheremisinoff, Paul N and Young, Richard Alan. Air Pollution Control and Design Handbook. s.l. : M Dekker, 1977. pp. 672-673. Vol. 2.
3. Urban Air Quality in Europe. Boulter, P G, Borken-Kleefeld, J and Ntziachristos, L. [ed.] M Vianna. Berlin Heidelberg : Springer-Verlag, 2013, Handbook of Environmental Chemistry, Vol. 26, pp. 31-54.
4. NOx emissions in China: historical trends and future perspectives. Zhao, B, et al. 13, 2013, Atmospheric Chemistry and Physics, pp. 9869-9897.
